Lights and shadows of implantable cardioverter defibrillators implantations in hypertrophic, dilated and arrhythmogenic right ventricular cardiomyopathies.
Posted: April 29, 2013 Filed under: Medical Devices, Opinions | Tags: bradycardia, defibrillator, eHealthcare, Health 2.0, ICD, implantable cardioverter defibrillators, lights, Medical Devices, pacemaker, SCD, sudden cardiac death Leave a commentIn the past decades, implantable cardioverter defibrillators (ICDs) have definitively proved to be superior to antiarrhythmic drugs for the prevention of arrhythmic sudden cardiac death (SCD). Several studies have proved this hypothesis both for primary or secondary (i.e. after a sustained ventricular tachycardia with hemodynamic compromise or ventricular fibrillation) prevention of SCD. However, some complications often occur with ICD implantation, which may be particularly serious for young individuals.
Indications for ICD implantation in young people are mostly ion channel diseases and cardiomyopathies. The latter are structural abnormalities of the myocardium, related to genetic abnormalities, predisposing to malignant ventricular arrhythmias and requiring ICD for the prevention of SCD in some cases.
Hypertrophic cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is one of the most common cardiomyopathies and occurs in the 0.2% of the general population1. It is characterized by localized asymmetrical hypertrophy of the heart and disarray of myocardial cells and filaments. This leads to impaired transmission of electrophysiologic impulses, with an unstable electrical substrate, potentially causing lethal ventricular tachyarrhythmias and SCD.
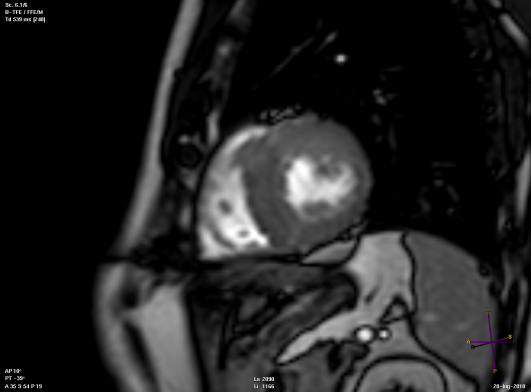
Short axis view of a cardiac magnetic resonance imaging of a patient affected by hypertrophic cardiomyopathy involving the interventricular septum and the anterior wall of the left ventricle.
ICD implantation in HCM: lights
A minority of patients with HCM are judged to be at increased risk for SCD, whose rate is estimated to be about 1% per year 2. However, malignant ventricular arrhythmias remains the most frequent cause of death in this population.
Beta-blockers have failed to demonstrate significant protection from SCD in HCM patients. Type I and III anti-arrhythmic drugs, including amiodarone, have been abandoned because of inefficacy and pro-arrhythmic side effects. Experiences with endocardial and epicardial mapping and ablation in HCM are poor and limited to highly selected patients.
ICDs, conversely, have been proved effective in terminating life-threatening ventricular tachyarrhythmias in HCM and are currently considered the only available tools altering the natural course of the disease and prolonging life.
Appropriate intervention rates of ICD in HCM have been esteemed to be about 11% and 4% for secondary and primary prevention respectively3.
Current recommendation for ICD in HCM
HCM has heterogeneous clinical manifestations in different individuals and a not well predictable clinical course. Targeting HCM-patients for prophylactic ICD implantation can therefore be challenging. However, some “risk factors” have been individuated, that commonly guide the decision for ICD implantation. Current international guidelines recommend ICD in all cases of secondary prevention or when familiar history of SCD, marked left ventricular (LV) hypertrophy or recent unexplained syncopal episodes are present. The role of ICD is uncertain for non-sustained VT or an abnormal blood pressure response with exercise.
ICD implantation in HCM: shadows
Previous studies have reported up to 5.1%/year rate of ICD-related complications in HCM4.
The most frequently reported is inappropriate shock delivery, mostly due to erroneous detection and treatment of atrial fibrillation by the ICD. A retrospective study on 334 consecutive HCM patients with an ICD, showed a rate of appropriate vs. inappropriate shock delivery of 2.3 and 4.6%/year5. Moreover, a recent meta-analysis involving 2190 ICD-recipients affected by HCM, showed a rate of appropriate vs. inappropriate ICD interventions of 3.3 vs. 4.8%/year respectively4.
Another extremely rare complication with ICDs in HCM, is represented by loss of capture due to high pacing thresholds (i.e. energy required for effective right ventricular pacing). This is related to ventricular hypertrophy and may be prevented by accurate individuation of the optimal pacing threshold during ICD programming.
Lengthened mitral valve leaflets, and left ventricular outflow tract obstruction due to LV hypertrophy, also confers a relatively higher risk of infection and endocarditis to HCM patients, which must be considered when an ICD implantation is programmed. Moreover, since ICD implantation in HCM is commonly performed at a relatively young age, patients are supposed to necessitate of a number of interventions for pulse generator substitution over their entire life. For this reason, risk of infection and endocarditis (related to ICD substitutions) is amplified. Eventual needing of ICD and intra-cardiac leads extraction over time must be considered. This is a high-risk procedure, possibly complicated by cardiac tamponade, shock, anemia, arrhythmias or even death.
Finally, ICD implantation can be accompanied by depression, anxiety, reduced quality of life, particularly in young people.
Idiopathic dilated cardiomyopathy
Idiopathic dilated cardiomyopathy (IDCM) is a myocardial disease characterized by LV dilation and systolic dysfunction, commonly resulting in heart failure (HF) and for whom an etiological basis cannot be identified. IDCM is relatively rare (36.5% new cases/year/100.000 persons) but accounts for nearly 10.000 deaths/year in the United States, both due to HF and arrhythmic SCD6.
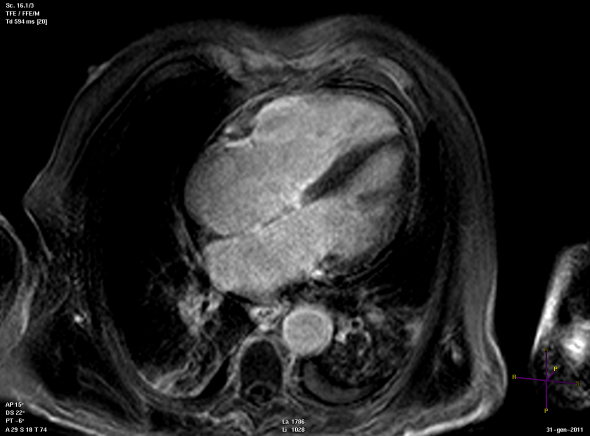
Long axis view of a cardiac magnetic resonance imaging of a patient affected by dilated cardiomyopathy
ICD implantation in IDCM: lights
ICD are effective for the prevention of SCD in IDCM and can favorably alter the natural course of the disease. Previous studies have showed a rate of appropriate interventions of 5 to 7.5%/year in IDCM-patients with an ICD implanted for primary prevention 7-8. A large meta-analysis has showed a reduction of mortality with ICD of about 3.5%/year compared to the best medical therapy in IDCM 9.
Recommendation for ICD in IDCM
The most important risk factor for ventricular arrhythmias in primary prevention of SCD in IDCM, is represented by severe contractile dysfunction, quantified by the measurement of the ejection fraction (EF) at echocardiography. Functional status, assessed by the New York Heart Association (NYHA) class, also plays a role.
Current international guidelines recommend ICD always for secondary prevention. In case of primary prevention, ICD must be implanted in patients with LVEF ≤35% and NYHA class II and III10. However, in a real-life setting, the decision making for patients with IDCM is more complex, because issues such as additional cardiac resynchronization therapy (CRT), co-morbidities, the potential to improve LVEF over time, and eventual genetic etiology also should be considered. With regard to the last issue, carriers of mutations in gene encoding for laminin, are tough to be at increased risk of complete atrio-ventricular block, malignant ventricular arrhythmias and SCD, and may therefore require ICD implantation independently from the EF%.
ICD implantation in IDCM: shadows and warnings
Some shadows obscure the scenario of SCD prevention with ICDs in IDCM.
An important issue regards the high number of patients to be treated in order to save one life because of a currently adopted risk stratification process that appears to lack specificity.
Another topic is the correct timing for ICD implantation for primary prevention. A post hoc analysis of the DEFINITE trials 11 showed that only patients who had received their ICD not later than 3 months after the diagnosis of IDCM would have benefit from implant. However, a significant number of IDCM-individuals show marked improvement of the EF over time, up to values higher than those for which ICD implantation is currently recommended. The goal therefore, appears to be an early and correct individuation of those subjects who will have a negative clinical course, and will therefore require an early ICD implantation for primary prevention.
Similarly to HCM, ICD-related complications have been also reported by various studies, among whom:
- inappropriate shocks, with consequent reduction of quality of life;
- infections;
- risks related to eventual lead extractions;
- depression and anxiety.
Arrhythmogenic right ventricular cardiomyopathy
Arrhythmogenic right ventricular cardiomyopathy (ARVC) is a genetic cardiomyopathy characterized by progressive fibro-fatty replacement of the right ventricular myocardium, sometimes extending also to the left ventricle. The clinical presentation is usually related to ventricular tachycardia (VT) with a left bundle branch block pattern or ventricular fibrillation (VF) leading to SCD. ARVC is a progressive disease ultimately leading to HF.
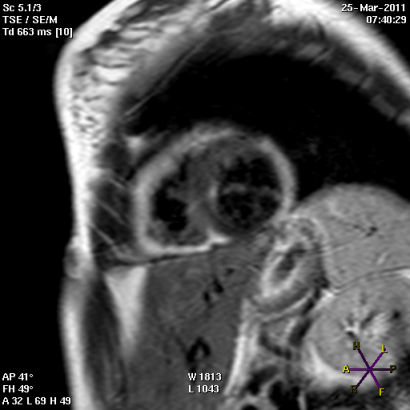
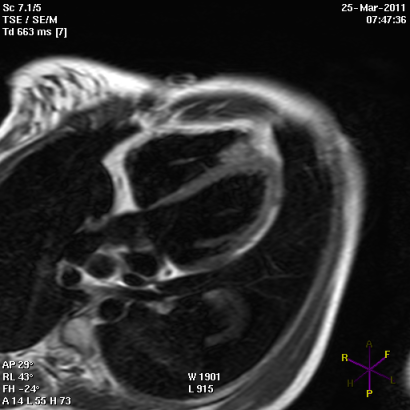
Panel A
Panel B
Short (Panel A) and long axis views (panel B) of a cardiac magnetic resonance imaging of a patient affected by arrhythmogenic right ventricular cardiomyopathy, with evident fibro-fatty replacement of the right ventricle wall, the apex and partial involvement of the left ventricle wall.
ICD implantation in ARVC: lights
The therapeutic options for ventricular arrhythmias in ARVC include catheter ablation and antiarrhythmic drugs such as beta blockers, sotalol and amiodarone. However, these strategies have proved to improve symptoms but not to increase survival. Nowadays, ICD remains the most effective safe-guard against SCD in ARVC.
In a large multicenter international study enrolling ARVC patients with an ICD implanted for primary prevention, appropriate device interventions were observed in one fourth of patients after 5 years. The annual rate of potentially “life-saving” shocks against VF was 3.3% and the estimated benefit of ICD implantation was of 23% after 2 years 12. Another recent prospective study enrolling a cohort of ARVC patients with an ICD implanted for primary prevention, reported appropriate device interventions in nearly one-half of individuals over a period of 4.7 years 13.
Recommendation for ICD in ARVC
As for IDCM and HCM, ICD implantation is recommended in ARVC for secondary prevention (aborted SCD, VF or hemodynamically unstable sustained VT). ICD is also recommended for primary prevention in patients with LVEF≤35%, severe right ventricular dilation and/or dysfunction, a syncopal episode suggestive for VT or VF or an affected family member with SCD. Other risk factors include: non-sustained VT, early onset of the disease and competitive sport activity.
ICD implantation in ARVC: shadows.
Several studies have proved a relatively high incidence of device-related complications in ARVC patients with an implanted ICD. In a study of 132 patients, five individuals required an additional lead because of pacing failure and one patient died from endocarditis secondary to device infection 14. Progression of fibro-fatty replacement of the myocardial tissue has been associated with high pacing thresholds and impedances and eventual loss of capture. Up to 37% lead-related complication in 7 years have been described in ARVC patients with an implanted ICD 15.
Considerations
The decision regarding ICD implantation is highly significant for any individual at risk of SCD. The clinical decision-making process itself is complex and imply consideration of a number of different aspects. Despite ICD is the only life-prolonging therapy in cardiomyopathies, eventually associated complications should be considered.
Modern technology, such as that utilized by NayaMed, has currently developed sophisticated algorithms allowing an accurate discrimination of supraventricular from ventricular arrhythmic episodes and properly guiding ICD therapies. A growing knowledge of those algorithms by the cardiologists would hopefully reduce the rate of inappropriately delivered shocks by ICDs.
Reliable RV lead alerts and RV lead integrity algorithms, also a constant automatic measurement of leads’ impedances and P, R Waves amplitude have also been implemented in the ICDs of NayaMed, preventing the problems caused by a lead failure. However, despite such algorithms, a complete abolition of ICD-related complications in cardiomyopathies is unreliable. Therefore, an accurate risk stratification appears necessary, as well as a careful weigh of the relative risks and benefits of ICD implantation in each individual.
Dr. Annamaria Martino
Dr. Leonardo Calo
Policlinico Casilino
Roma, Italy
References
1) Gersh J, Maron BJ, Bonow RO et al. 2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy: A Report of the American College of Cardiology Foundation/ American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2011; 58:e212-260
2) Maron BJ. Hypertrophic cardiomyopathy: a systematic review. JAMA 2002; 287:1308-1320
3) Maron BJ, Spirito B, Shen WK et al. Implantable cardioverter defibrillators and prevention of sudden cardiac death in hypertrophyc cardiomyopathy. JAMA 2007; 298: 405-412
4) O’Mahony C, Lambiase PD, Quarta G et al. The long-term survival and the risk and benefits of implantable cardioverter defibrillators in patients with hypertrophic cardiomyopathy. Heart 2012;98:116-125
5) Schinkel AF, Vriesendorp PA, Sijbrands EJ et al. Outcome and complications after implantable cardioverter defibrillator therapy in hypertrophic cardiomyopathy: systematic review and meta-analysis. Circ Heart Fail 2012;5:552-559
6) Codd MB, Sugrue DD, Gersh BJ et al. Epidemiology of idiopathic dilated and hypertrophic cardiomyopathy. A population based study in Olmsted County, Minnesota, 1975–1984. Circulation 1989;80:564–72
7) Bardy GH, Lee KL, Mark DB, Poole JE et al. Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) Investigators. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 2005;352:225–237.
8) Kadish A, Dyer A, Daubert JP et al; Defibrillators in non-ischemic Cardiomyopathy Treatment Evaluation (DEFINITE) Investigators. Prophylactic defibrillator implantation in patients with nonischemic dilated cardiomyopathy. N Engl J Med 2004;350:2151–2158.
9) Desai AS, Fang JC, Maisel WH, Baughman KL. Implantable defibrillators for the prevention of mortality in patients with nonischemic cardiomyopathy: a meta-analysis of randomized controlled trial. JAMA 2004;292: 2874–287
10) Zipes DP; Camm AJ, Borggrefe M et al. ACC/AHA/ESC 2006. Guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Europace 2006; 8:746-837
11) Kadish A, Schaechter A, Subacius H et al. Patients with recently diagnosed nonischemic cardiomyopathy benefit from implantable cardioverter defibrillators. J Am Coll Cardiol 2006;47:2477–2482
12) Corrado D et al. Prophylactic Implantable Defibrillator in Patients With Arrhythmogenic Right Ventricular Cardiomyopathy/Dysplasia and No Prior Ventricular Fibrillation or Sustained Ventricular Tachycardia. Circulation. 2010;122:1144-1152.
13) Bhonsale A, James CA, Tichnell C et al. Incidence and predictors of implantable cardioverter-defibrillator therapy in patients with arrhythmogenic right ventricular dysplasia/ cardiomyopathy undergoing implantable cardioverter-defibrillator implantation for primary prevention. J Am Coll Cardiol, 2011;58:1485-96
14) Corrado D, Leoni L, Link MS et al. Implantable cardioverter-defibrillator therapy for prevention of sudden death in patients with arrhythmogenic right ventricular cardiomyopathy/dysplasia. Circulation 2003;108:3084–91.
15) Wichter T, Paul M,Wollmann C et al. Implantable cardioverter/defibrillator therapy in arrhythmogenic right ventricular cardiomyopathy. Single-center experience of long-term follow-up and complications in 60 patients. Circulation 2004;109:1503–8.
Thank you for visiting us at the ESC Congress 2012 in Munich
Posted: September 10, 2012 Filed under: NayaMed spirit | Tags: bradycardia, cardiac electrophysiology, cardiac techniques, congress, eHealthcare, ESC, Germany, Health 2.0, healthcare organizations, ICD, Medical Devices, Munich, pacemaker, pacemakers, video 4 CommentsNayaMed products in a few words
Posted: August 20, 2012 Filed under: Medical Devices | Tags: bradycardia, cardiac, cardiac electrophysiology, cardiac techniques, cardiology, conventional devices, defibrillator, eHealthcare, Health 2.0, ICD, Medical Devices, pacemaker, pacemakers, St Jude Medical, technical advisor Leave a commentAs a new appearance on the CRDM Market, customers often ask: “Who are you NayaMed?”, “What are your products and why should we chose you for our patients?”
Well, mostly because we offer a simple online platform with a permanent access to your products, giving full transparency and allowing permanent access to ordering, digital inventory, stock management access and technical support.
Also, because as a clinician, you get the product you need when you need. A product that has been designed and engineered to perfectly match the NayaMed philosophy – Smart and Simple.
We wanted the clinicians to be comfortable when using our devices, so we asked our Technical Advisors to present the NayaMed products in just a few minutes.
Mary Soranno and Marina Mancusi, our technical advisors for the UK and the Italian market did a wonderful job and we are inviting you to see a very brief, but complete presentation of our products in English or Italian.
English Version:
Italian Version:
NayaMed devices made simple. Watch it now!
Posted: August 13, 2012 Filed under: Medical Devices, Uncategorized | Tags: bradycardia, cardiac, cardiac electrophysiology, cardiac techniques, cardiology, conventional devices, defibrillator, eHealthcare, Health 2.0, healthcare applications, heart failure, ICD, Medical Devices, pacemaker, pacemakers, video Leave a commentMeet Mary Soranno, our technical advisor for the UK market presenting all the features of the NayaMed pacemakers & defibrillators. (Better than a long blog post or brochure!).
First NayaMed ICD implant performed only with remote support
Posted: February 2, 2012 Filed under: Medical Devices, NayaMed spirit | Tags: conventional devices, Health 2.0, ICD, innovation, Medical Devices, Remote Support Leave a comment “I know that the patient is ready for the implant, but I’m late because of the snow, I’m so sorry!”;
“I know that the patient is ready for the implant, but I’m late because of the snow, I’m so sorry!”;
“Hello Dr.Y, I’ll do my best to be here as soon as possible for the first scheduled procedure but I’m afraid that I won’t be able to arrive before one hour”…
How many times the “traditional” technical advisors have already had to face such embarrassing situations? And how many times do the physicians have to change the EP lab program because they need a technical support? Really often I can say…but nor today and in the future with NayaMed!
Today at one of our customer in Italy, a NayaMed implant was performed ONLY with a remote support, without anyrepresentative in the field!!!
At the beginning of the procedure, I was in contact with the nurse online. She had performed all the initial tests on the ICD autonomously, just looking at NayaMed ICD Job aids and she asked me only few suggestions for the procedure.
At the end of the implant, the nurse called me back and we discussed together with the physician about how to optimize the parameters for our patient. It was a great and efficient work: I was available for them despite the snow at their time, even after some last-minute changes in their activity in the EP lab. Everything was done exactly in the NayaMed spirit: leading our customers to become autonomous in implanting conventional devices, being aware of having an Online Technical Support that is always available and ready for them, anytime, anywhere.
This is really a new frontier of smart and simple healthcare isn’t it?
Marina Mancusi
NayaMed Technical Advisor
Kodak, the Roman Empire, Italian unity and Healthcare
Posted: January 19, 2012 Filed under: Medical Devices, Opinions | Tags: healthcare applications, healthcare organizations, Medical Devices 1 Comment It’s an odd title, I know, but please bear with me; there’s a reason for that.
It’s an odd title, I know, but please bear with me; there’s a reason for that.
Eastman Kodak Company (commonly known as Kodak) was founded in 1892 in in Rochester, New York. It is a company that I find very interesting because in the late 1800 Kodak innovated profoundly the photographic business by selling inexpensive cameras and making large margins on consumables[1]. Kodak strategy was so successful that in the late 1976 it was leading the US market with over 90% market share in film sales and 85% in camera sales. Astonishing, isn’t it?
So what happened? Why all people I know look at Kodak today as a modern antique?
I believe that the answer is in a study made in 2005 by the Harvard Business School, according to which Kodak failed because its “seemingly unassailable competitive positions fostered Kodak’s unimaginative executive culture”[2]
Is that correct? Is it all about imagination? I believe so, well maybe not just imagination but also being able to challenge the status quo to face future challenges. I believe that empires, companies and cultures, at a certain point of their lifecycle start to suffer of what I call “The Roman Empire Syndrome”.
You might say: “Hold on a second. What is the link between a global company selling camera films and the Roman Empire?”
We all know the history of the Roman Empire, its success and continuous growth over several hundreds of years. In the third century Rome faced a number of crises, but the Empire was too vast and ungovernable, therefore unable to react quickly to new challenges. That was the beginning of the end, started in 293 AD, when the emperor Diocletian decided to split the empire first in two parts and then in four transforming it into a tetrarchy.
So what I believe is that intrinsically big organizations contain the seed of their own end; for the simple reason that when they become very large they also become too innerly focused and unable to adapt quickly to a changing environment[3].
Coming back to today, we are facing the biggest economic challenge of the European Union. The current situation is generating additional taxes and austerity measures, including the healthcare sector which has more and more restricted budgets. On the other hand, the European population is growing older and older. If we only consider the five biggest European countries (Germany, UK, France, Italy and Spain) 18% or more of the population is above 65, with Italy and Germany being more than 20%[4].
So we could say that the European healthcare system (including National Health, Industry, Hospitals, etc…) is facing a major challenge: how can I treat an increasing patient population, demanding always better quality of care, with flat or shrinking budgets?
This challenge is addressed to all actors: governments, institutions, hospitals and also the Industry. I think that NayaMed is nothing else than one of the first answers (and probably not the only one) to the real needs of Healthcare today. I think that initiatives like NayaMed will sparkle and inspire the medical device industry in how we can achieve more with less. Because, similarly to the Roman Empire or Kodak, I don’t see today the major medical device companies taking a step-change and rethinking their business model, adapting to the changing environment[5].
Massimo D’Azeglio, an Italian politician and patriot, said during the Italian Unification: “We have made Italy, now we must make the Italians”[6]. Today, after having spent a lot of efforts in building NayaMed, it is exactly the same challenge that we are facing. We have generated a new sparkle in the healthcare market, now we have to ignite the change in our customers, partners and even competitors.
So, to make a long story short, yesterday NayaMed did the first ICD implant in the hospital S.Orsola in Bologna… and today I’m feeling like Johnny Utah in Point Break when, just before parachuting down from a plane for the first time, one of the other surfers tells him “You’re about to jump out a perfectly good airplane Jonny, how do you feel about that?”[7].
My answer would be: good.
Andrea Pedrazzini
Marketing Manager NayaMed
[1] Business model that will be readopted in the future by the home printing market and personal shaving market
[2] Gavetti, G., Henderson, R. and Giorgi, S., Kodak and the Digital Revolution (A), 2005, Harvard Business School, HBS Press
[3] We could talk about Darwinism also, but someone else already applied the principles of the Natural Selection to companies better than me
[4] CIA World Fact book
[5] I particularly refer to the European Market and the European healthcare
[6] I know, the quote is controversial. Apparently what Massimo D’Azeglio said was: “Unfortunately we have made Italy but we haven’t made Italians”. According to the historians Simonetta Soldani and Gabriele Turi, the sentence that I’m quoting was coined in 1886 by Ferdinando Martini “in an attempt to translate the political sense” of the original one.
[7] Point Break – 1991. My wife, which is a way better English speaker than I am, suggest that the sentence should be “You’re about to jump off a perfectly good airplane Jonny, how do you feel about that?”. I report the sentence as in the movie, acknowledging that a bank-robber-surfer-that-is-jumping-off-a-plane might do an English mistake…
How to continue to deliver high-quality healthcare products when rising life expectancy is straining health systems across Europe?
Posted: January 11, 2012 Filed under: eHealthCare, Infographic, Medical Devices, Video | Tags: Health 2.0, infographic, me, Medical Devices, video Leave a commentAcross Europe, the continuous increase in life expectancy is resulting in an ever larger ageing population. Although this is should be seen as very positive, it does raise some concerns in relation to all European healthcare systems. Indeed, as the population ages the risk of chronic cardiac disease increases (such as the coronary artery disease bradycardia), and healthcare systems struggle to face this increasing demand due to flat or decreasing budgets.
Notwithstanding this budgetary pressure, medical treatment is continually progressing by offering more advanced and more expensive therapies. Today’s healthcare environment sees very few medical innovations that are targeted towards reducing costs for treating specific diseases.
As with most successful innovations over recent years, real advantages do not arise from lifting product features to the functional maximum, but from combining existing technologies and services to create smart and sustainable solutions for all relevant players in the system. NayaMed represents such a solution which goes beyond outdated categories such as “the best features” or “the cheapest price”.
A new tool to help the patients with moderate to severe Heart Failure
Posted: December 8, 2011 Filed under: Medical Devices | Tags: bradycardia, cardiac, cardiology, heart failure, Medical Devices, pacemakers Leave a commentRecentl y, an Australian company has developed a new heart assisting device designed to help patients suffering of Heart Failure and with mild to moderate symptoms (NYHA II – IV, Stage C).
y, an Australian company has developed a new heart assisting device designed to help patients suffering of Heart Failure and with mild to moderate symptoms (NYHA II – IV, Stage C).
The system represents a balloon that is surrounding the aorta and it inflates and deflates in the same rhythm with the heart contraction. The idea is a new tentative in addressing the heart failure patients that despite optimal drug and device treatment, they still have a poor heart function and significant HF symptoms (shortens of breath, tiredness with moderate activity).
What is interesting is the device tries to improve the cardiac output and also 2 important hemodynamic factors; decrease afterload and recovering a better contractility by a better perfusion of the coronaries. The contractility is an indirect improvement but we could assume that a better perfused myocardium is also contracting better.
In a normal person, the coronary arteries are perfused during diastole. A heart failure patient usually has a long contraction time of the myocardium during the heart cycle which leaves less time for the diastole and therefore less time to perfuse the coronaries.
The apparatus is currently studied in some hospitals in the United States under the FDA guidelines. It consists of a cuff that wraps around the ascending aorta and is connected to a pump by a tube that passes through the skin. This pump is external to the body and it moves air in and out of the cuff. A pacemaker lead is fixated to the heart to detect the appropriate timing when the balloon has to be inflated or deflated. When this one inflates it helps push the blood to the body and to the coronaries. During deflation, the workload or pumping required by the left heart is reduced.
Another notable aspect is that although the device needs heart surgery to be implanted it doesn’t come in direct contact with the blood flow and there is no major incision on the heart except the placement of the pacemaker lead which is minimal.
The constraint that we can think of this tool is that, being external; it has to be permanently carried by the patient in order for this one to take advantage of its benefits. For some activities, like showering the device can be disconnected but not more than 15 minutes, as the manufacturer recommends.
The current clinical results are promising; significant improvements in NYHA Class reduction, Quality of Life, and Left Ventricular Ejection Fraction. This makes the C-Pulse® a possible new treatment for the Heart Failure patients.
We think about the cardiac pacemakers implanted in the fifties that that were initially external and now are implantable and slightly larger than a coin. Maybe in the future this entire new heart assisting system could be minimized and placed completely under the skin.
References:
http://www.sunshineheart.com consulted on the 4th of December 18:00 GMT+1
http://medgadget.com/2011/11/sunshine-hearts-c-pulse-cardiac-pump-proves-itself-in-clinical-trial.html consulted on the 4th of December 18:00 GMT+1
http://www.marketwatch.com/story/sunshine-heart-announces-results-of-its-c-pulser-feasibility-trial-at-the-transcatheter-cardiovascular-therapeutics-conference-2011-11-07 consulted on the 4th of December 18:00 GMT+1
Alexandru Trif
Product Marketing Manager
Job Offer: TECHNICAL ADVISOR GERMANY
Posted: December 2, 2011 Filed under: Job Posts | Tags: eHealthcare, Germany, Job post, Medical Devices, technical advisor Leave a comment Lausanne – Switzerland – Permanent position
Lausanne – Switzerland – Permanent position
NayaMed is looking for a regional Technical Advisor. This role entails assisting electrophysiologists, cardiologists and clinicians in managing the programming of medical devices (pacemakers and defibrillators). As part of a centralized Care Center Unit, the Technical Advisor will work at the company’s headquarters.
Job description:
– Provide remote training of hospital staff on the medical device technologies
– Advise clinicians on device programming during implants, follow-up visits and troubleshooting situations
– Answer customers’ queries regarding technical issues, rapidly and precisely
– Help customers in managing product stock at hospital level using internal Inventory Management System
– Proactively contact the customers for training and helping them with new or changed features
– Play active role in the customer experience and collect info appropriately in the internal CRM system
– Support the sales process in relation to technical and administrative issues
– Animate community initiatives as an expert on the products
– Collaborate with the marketing team in order to develop new products and solutions, optimize processes at customer level
– Actively participate in internal and external education with technical and clinical literature
Who we are looking for:
– Higher education or degree in engineering, biomedical engineering degree preferred
– Other relevant background or education can be medical or paramedical
– Experience in medical devices, experience as a Technical Consultant in Cardiac Rhythm Management is a plus
– Spontaneous and friendly personality, service-minded
– Must be able to work well within a multicultural environment
– Must have excellent communication, interpersonal and computer skills
– Should be able to ameliorate customer service and all the staff’s skills
– Must be able to manage, use, and populate a knowledge database
– Good technology skills in relation with web support and unified communication
– Must be able to adapt well to change, enjoy working in a fast-paced environment
– Capacity and willingness to learn fast
– Should derive energy from helping others
– Fluency in German is mandatory; English and French would be seen as strong assets
If you are interested, please send your CV and cover letter to steve.forrer@nayamed.com